Post exercise vasovagal syncope
CTP Krediet *, AAM Wilde §, JR Halliwill + W Wieling *
Departments of *Internal Medicine and §Cardiology (AAMW), Academic Medical Center/ University of Amsterdam, The Netherlands; + Department of Human Physiology, University of Oregon, Eugene, United States of America
A 28 years old male fire fighter with an excellent general condition lost transiently consciousness after termination of a routine check-up exercise test [1]
. The episode occurred, while he was sitting motionless on the bicycle ergometer after the test, and his skin fold thickness was being measured. The patient was declared unfit for his job. The patient was referred to our syncope unit for further analysis of the episode.
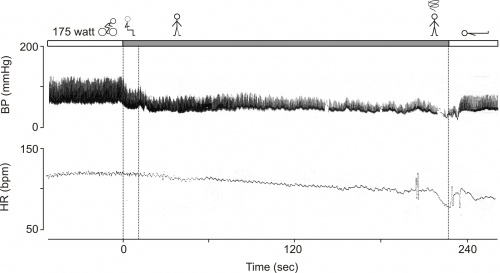
The exercise test was repeated using continuous non-invasive blood pressure monitoring. The figure shows the original continuous blood pressure and instantaneous heart rate tracing.
After cycling at 175 watts, the subject was asked to sit upright briefly and then stand for several minutes. As can be seen blood pressure and heart rate declined over several minutes in the upright position, leading to an eventual post-exertion vasovagal response with obvious bradycardia and hypotension. The subject was then placed in the supine position, and subsequently pressure recovered quickly. The patient recognized the symptoms. The exercise test was repeated once more. This time the patient was told to “keep moving” after the test and syncope did not occur. Post-exercise vasovagal syncope was diagnosed. The patient was reassured regarding the nature of his problem and was advised to avoid standing motionless after heavy exercise. Since his episodes occurred only during a predictable provocation i.e. motionless sitting/standing and he experienced clear preceding symptoms,. we declared the patient fit for work as a fire fighter. During a 5 year follow-up period, he has had no episodes of syncope.
Editor's Comments
The majority of cases of syncope occurring after exercise fall into two related categories: 1) an exaggerated form of the normal post-exercise hypotension response, and 2) neurally mediated syncope (i.e., vasovagal reactions). It is probable that the former triggers the latter. [1]
Post-exercise hypotension
During post-exercise hypotension, the mechanisms that regulate arterial pressure drive a modest but sustained reduction in arterial pressure. The cause of PEH is two-fold, involving both neural and local vascular mechanisms [2] [3]. In comparison to rest, PEH is characterized by a persistent drop in systemic vascular resistance that is not completely offset by increases in cardiac output [2][3]. The vasodilatation that underlies PEH is not restricted to the sites of active skeletal muscles, but involves inactive regions as well. The associated rise in arterial blood inflow through the vasodilated regions contributes to an increase in venous pooling of blood.
During exercise, rhythmically contracting skeletal muscles in the lower part of the body reduce the degree of venous pooling by squeezing veins, in effect, pumping blood back to the heart. This phenomenon is known as the ‘muscle pump’. This “pump” is absent during passive recovery from exercise. The increase in venous pooling, in conjunction with the loss of plasma volume associated with exercise, leads to a reduction in central venous pressure (~ 2 mmHg supine) and cardiac filling [3][4]. Despite this fall in cardiac preload, stroke volume is maintained due to the reduction in cardiac after-load and a probable increase in cardiac contractility [2][4]. The net result of these influences on the blood vessels and heart is that cardiac output is elevated (heart rate is higher with unchanged stroke volume). Thus PEH is due to a persistent drop in systemic vascular resistance that is not completely offset by increases in cardiac output.
The main point is that PEH is common after moderate-intensity dynamic exercise. In general, this is a benign process and in the majority of individuals PEH is insufficient to cause syncopal symptoms.
The magnitude of PEH can be exaggerated in the seated or standing positions (compared to supine) [2][3][4][5] and arterial pressure may fall to the level at which pre-syncopal signs or symptoms occur.
Neurally mediated syncope after exercise
It appears that the underlying mechanism for neurally mediated syncope occurring after the termination of exercise are similar to that of PEH. The sudden removal of the muscle pump activity, decreasing cardiac preload, may be a trigger, along with a rapid return of vagal tone to the heart at the termination of exercise. [1]
Characteristically, these events occur while the individual is standing motionless during the first 5 to 10 min after exercise [5][6]. Individuals rapidly recover in the supine position. It has been estimated that the incidence rate of neurally mediated syncope after routine treadmill testing may be in the order of 0.3-3 % [7]. However, when treadmill testing is immediately followed by passive head-up tilt testing, this percentage can increase up to 50-70% [7][8]. Neurally mediated syncope after exercise is considered to be a benign occurrence [1].
In a minute minority of cases of syncope during (exertional) exercise the vasovagal reaction is thought to be the underlying pathophysiological mechanism [1]. How the reaction in this circumstance is triggered remains to be elucidated [1][9]. Nausea prior to such episode is a commonly reported symptom. The following mechanisms may be involved.
- Supra-physiological stimulation of ventricular mechanoreceptors in the left ventricle, especially when cardiac filling is decreased at a high heart rate is thought to be a trigger for vasovagal reactions (i.e. the Bezold-Jarisch hypothesis).[10] Abrahamsen and Thoren made the observation that such stimulation leads also to reflex gastric dilatation and eventual vomiting in the cat.[11] However many dispute the Bezold-Jarisch hypothesis because evidence for the existence of ventricular mechanoreceptors in man is lacking.[10] The newly discovered mechano receptors in the coronaries of the dog could provide an alternative explanation for the results that once were thought to demonstrate the existence of the ventricular mechanoreceptors.[12]
- Holmqvist et al showed, using pancreatic polypeptide as a measure for vagal activity in man that abdominal vagal activity increases during maximal exercise, reaching its highest values after exertion.[13] They suggest that such high vagal outflow may also cause nausea.
Although patients suffering from “vasovagal syncope during exercise” have been studied intensely, there are no objectified observations of such loss of consciousness during exercise. This raises the question whether this disease as a separate entity truly exists. If not, this form of syncope could actually be the same as vasovagal occurring after exercise, only differing in time between time between stopping and loss of consciousness. In cases where this comes within a certain amount of time patients will be inclined to report that the lost consciousness during exercise, where in fact it happened shortly after. The importance of detailed history taking is also in these cases beyond doubt.
Syncope related to exercise may be the first indication of a dangerous underlying cardiovascular condition. First of all the diagnostic workup of patients presenting with exercise-related syncope is aimed at excluding these.
References
-
Parts of this case were published earlier: Krediet CT, Wilde AA, Wieling W, Halliwill JR. Excercise related syncope: when it’s not the heart [review]. Clin Auton Res 2004; 14 Suppl 1:25-36. We thank Springer Verlag (Darmstadt, Germany) for permitting this reproduction.
- Kenney MJ and Seals DR. Postexercise hypotension. Key features, mechanisms, and clinical significance. Hypertension. 1993 Nov;22(5):653-64. DOI:10.1161/01.hyp.22.5.653 |
- Halliwill JR. Mechanisms and clinical implications of post-exercise hypotension in humans. Exerc Sport Sci Rev. 2001 Apr;29(2):65-70. DOI:10.1097/00003677-200104000-00005 |
- Halliwill JR, Minson CT, and Joyner MJ. Effect of systemic nitric oxide synthase inhibition on postexercise hypotension in humans. J Appl Physiol (1985). 2000 Nov;89(5):1830-6. DOI:10.1152/jappl.2000.89.5.1830 |
- Bjurstedt H, Rosenhamer G, Balldin U, and Katkov V. Orthostatic reactions during recovery from exhaustive exercise of short duration. Acta Physiol Scand. 1983 Sep;119(1):25-31. DOI:10.1111/j.1748-1716.1983.tb07301.x |
- Tsutsumi E and Hara H. Syncope after running. Br Med J. 1979 Dec 8;2(6203):1480. DOI:10.1136/bmj.2.6203.1480 |
- Holtzhausen LM and Noakes TD. The prevalence and significance of post-exercise (postural) hypotension in ultramarathon runners. Med Sci Sports Exerc. 1995 Dec;27(12):1595-601.
- Sakaguchi S, Shultz JJ, Remole SC, Adler SW, Lurie KG, and Benditt DG. Syncope associated with exercise, a manifestation of neurally mediated syncope. Am J Cardiol. 1995 Mar 1;75(7):476-81. DOI:10.1016/s0002-9149(99)80584-0 |
- Krediet CT, Wilde AA, Halliwill JR, and Wieling W. Syncope during exercise, documented with continuous blood pressure monitoring during ergometer testing. Clin Auton Res. 2005 Feb;15(1):59-62. DOI:10.1007/s10286-005-0241-3 |
- Hainsworth R. Syncope: what is the trigger?. Heart. 2003 Feb;89(2):123-4. DOI:10.1136/heart.89.2.123 |
- Abrahamsson H and Thorén P. Vomiting and reflex vagal relaxation of the stomach elicited from heart receptors in the cat. Acta Physiol Scand. 1973 Aug;88(4):433-9. DOI:10.1111/j.1748-1716.1973.tb05472.x |
- Wright CI, Drinkhill MJ, and Hainsworth R. Responses to stimulation of coronary and carotid baroreceptors and the coronary chemoreflex at different ventricular distending pressures in anaesthetised dogs. Exp Physiol. 2001 May;86(3):381-90. DOI:10.1113/eph8602208 |
- Holmqvist N, Secher NH, Sander-Jensen K, Knigge U, Warberg J, and Schwartz TW. Sympathoadrenal and parasympathetic responses to exercise. J Sports Sci. 1986 Autumn;4(2):123-8. DOI:10.1080/02640418608732108 |